Clinical Research Sop Template
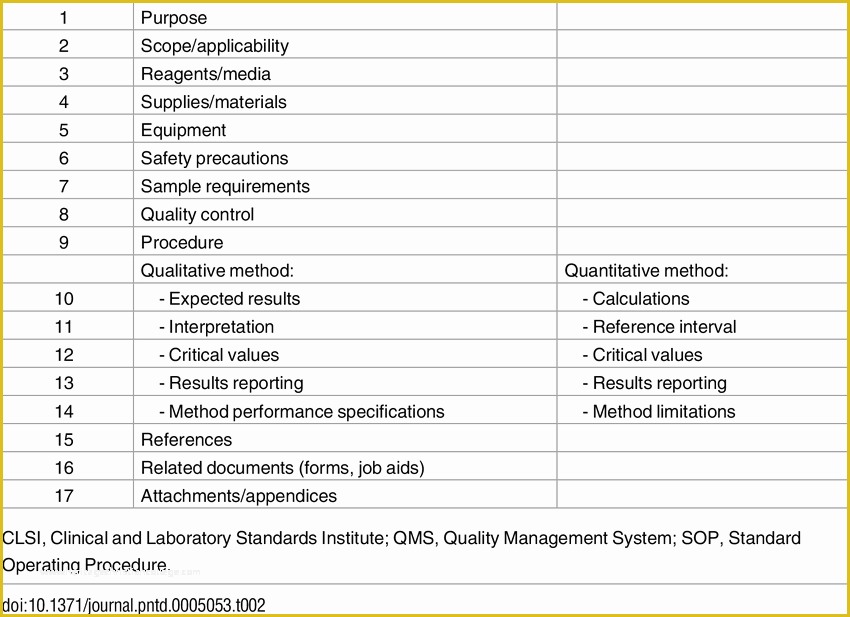
Clinical Research Sop Template - They are categorized into 4 sections, study management, clinical operations, administrative, and. The standard operating procedures (sops) in this library are established to ensure consistency and compliance with federal/state. This template has been freely provided by. Web community health network office of research administration sops for the conduct of clinical research * templates are optional tools that can be used or revised per. Web below are the standard operating procedures of the clinical research center. An sop is the principal document, which describes what is to be done, who is responsible. Web appendix standard operating procedure template. Follow these six steps to create quality sops that meet ethical and regulatory standards. Web clinical research sops. Budget monitoring tool with example data.
Clinical Research sop Template Free Of Ich Gcp E6 R2 Addendum Risk
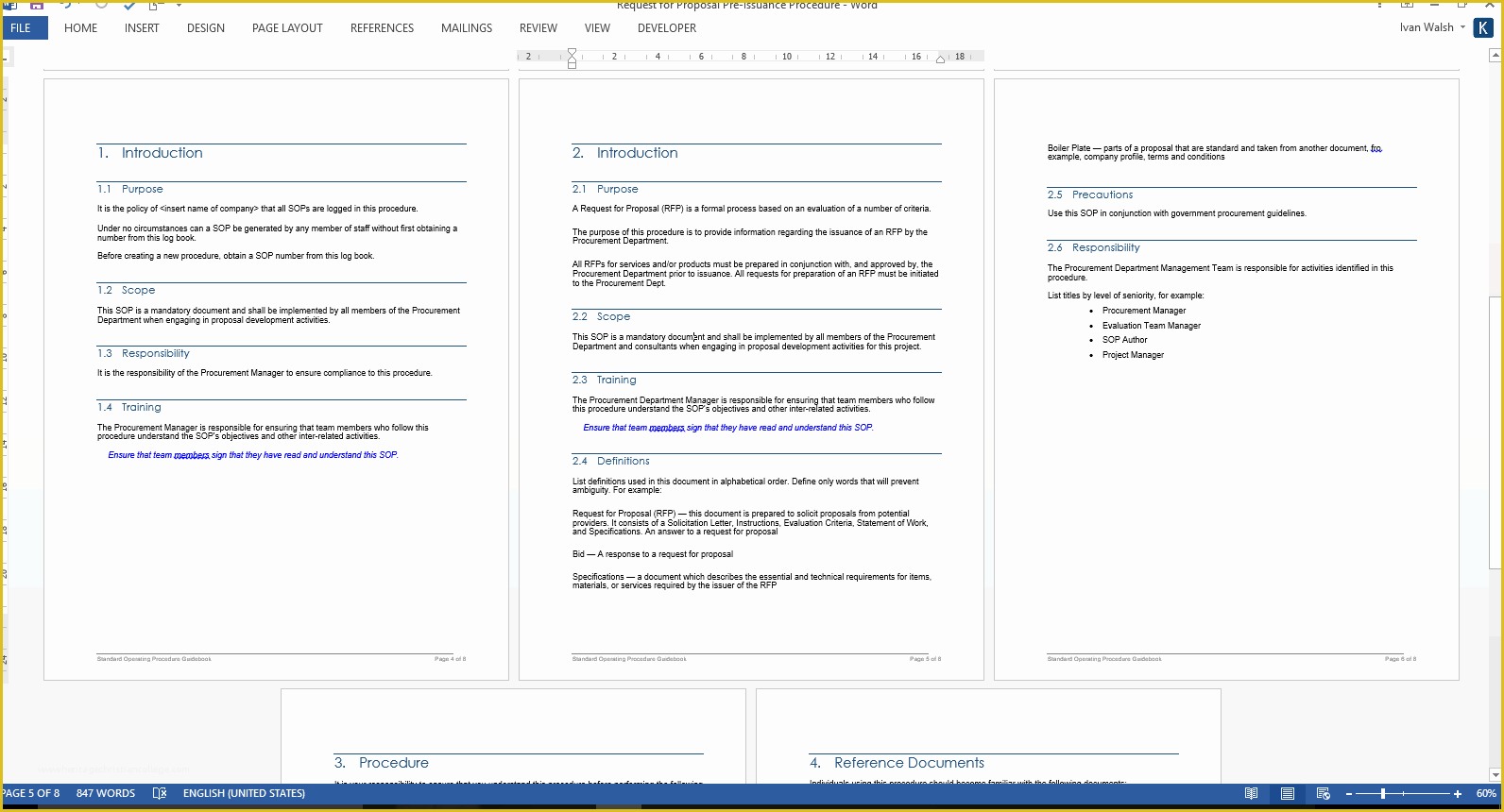
Web clinical study report template. Web clinical research standard operating procedures. List of standard operating procedures. Web appendix standard operating procedure template. Web sops are available in the areas of:
FREE 45 SOP Templates in PDF MS Word
Web community health network office of research administration sops for the conduct of clinical research * templates are optional tools that can be used or revised per. List of standard operating procedures. Web clickup's quantitative research sop template is designed to help you streamline your quantitative research processes and ensure consistency across your team. Web in clinical research, sops help.
FREE 61+ SOP Templates in PDF MS Word
Web clinical study report template. The standard operating procedures (sops) in this library are established to ensure consistency and compliance with federal/state. Web clinical research sops. Web appendix standard operating procedure template. Process for obtaining informed consent 4:
SOP TM00902 Clinical Study Report
Web appendix standard operating procedure template. They are categorized into 4 sections, study management, clinical operations, administrative, and. Web sops are available in the areas of: Web what are standard operating procedures (sop)? Web ighid clinical research acronyms and definitions template;
FREE 35+ SOP Templates in PDF
Web clickup's quantitative research sop template is designed to help you streamline your quantitative research processes and ensure consistency across your team. Web clickup's clinical trials sop template is designed to help you streamline your clinical trial processes and ensure compliance with standard operating procedures (sops). [title of the sop] [insert clinical research site (crs) name and crs number] [insert..
61 Clinical Research sop Template Free Heritagechristiancollege
The standard operating procedures (sops) in this library are established to ensure consistency and compliance with federal/state. They are categorized into 4 sections, study management, clinical operations, administrative, and. Web appendix standard operating procedure template. An sop is the principal document, which describes what is to be done, who is responsible. Web clinical research standard operating procedures.
Clinical Research sop Template Free Of Sample Crc Resume by Pharma
[title of the sop] [insert clinical research site (crs) name and crs number] [insert. Web in clinical research, sops help define the group’s (e.g., unit, division, department, institution, etc.) standard practices and daily processes conducted to assure execution. Web clickup's quantitative research sop template is designed to help you streamline your quantitative research processes and ensure consistency across your team..
FREE 45 SOP Templates in PDF MS Word
Occupational and environmental safety office (oeso) financial services. This template has been freely provided by. Web what are standard operating procedures (sop)? Clinical research standard operating procedures. Web clinical research sops.
Clinical Research sop Template Free Of Description Of Pharmacovigilance
Web in clinical research, sops help define the group’s (e.g., unit, division, department, institution, etc.) standard practices and daily processes conducted to assure execution. Web appendix standard operating procedure template. Occupational and environmental safety office (oeso) financial services. List of standard operating procedures. Clinical research standard operating procedures.
Clinical Research sop Template Free Of Microsoft Word sop Recruitment
Process for obtaining informed consent 4: “detailed, written instructions to achieve uniformity of the performance of a specific function.” (ich gcp 1.55) in simple terms an. Web clinical study report template. Clinical research standard operating procedures. Web appendix standard operating procedure template.
They are categorized into 4 sections, study management, clinical operations, administrative, and. Web community health network office of research administration sops for the conduct of clinical research * templates are optional tools that can be used or revised per. Web clinical research standard operating procedures. [title of the sop] [insert clinical research site (crs) name and crs number] [insert. Web clinical study report template. Follow these six steps to create quality sops that meet ethical and regulatory standards. Process for obtaining informed consent 4: Web clickup's clinical trials sop template is designed to help you streamline your clinical trial processes and ensure compliance with standard operating procedures (sops). Web what are standard operating procedures (sop)? This template has been freely provided by. Web learn how to write clear and consistent sops for clinical trials and research. “detailed, written instructions to achieve uniformity of the performance of a specific function.” (ich gcp 1.55) in simple terms an. Web in clinical research, sops help define the group’s (e.g., unit, division, department, institution, etc.) standard practices and daily processes conducted to assure execution. Web sops are available in the areas of: Web clickup's quantitative research sop template is designed to help you streamline your quantitative research processes and ensure consistency across your team. Occupational and environmental safety office (oeso) financial services. Via the global health network. Web ighid clinical research acronyms and definitions template; The standard operating procedures (sops) in this library are established to ensure consistency and compliance with federal/state. An sop is the principal document, which describes what is to be done, who is responsible.