Device History Record Template
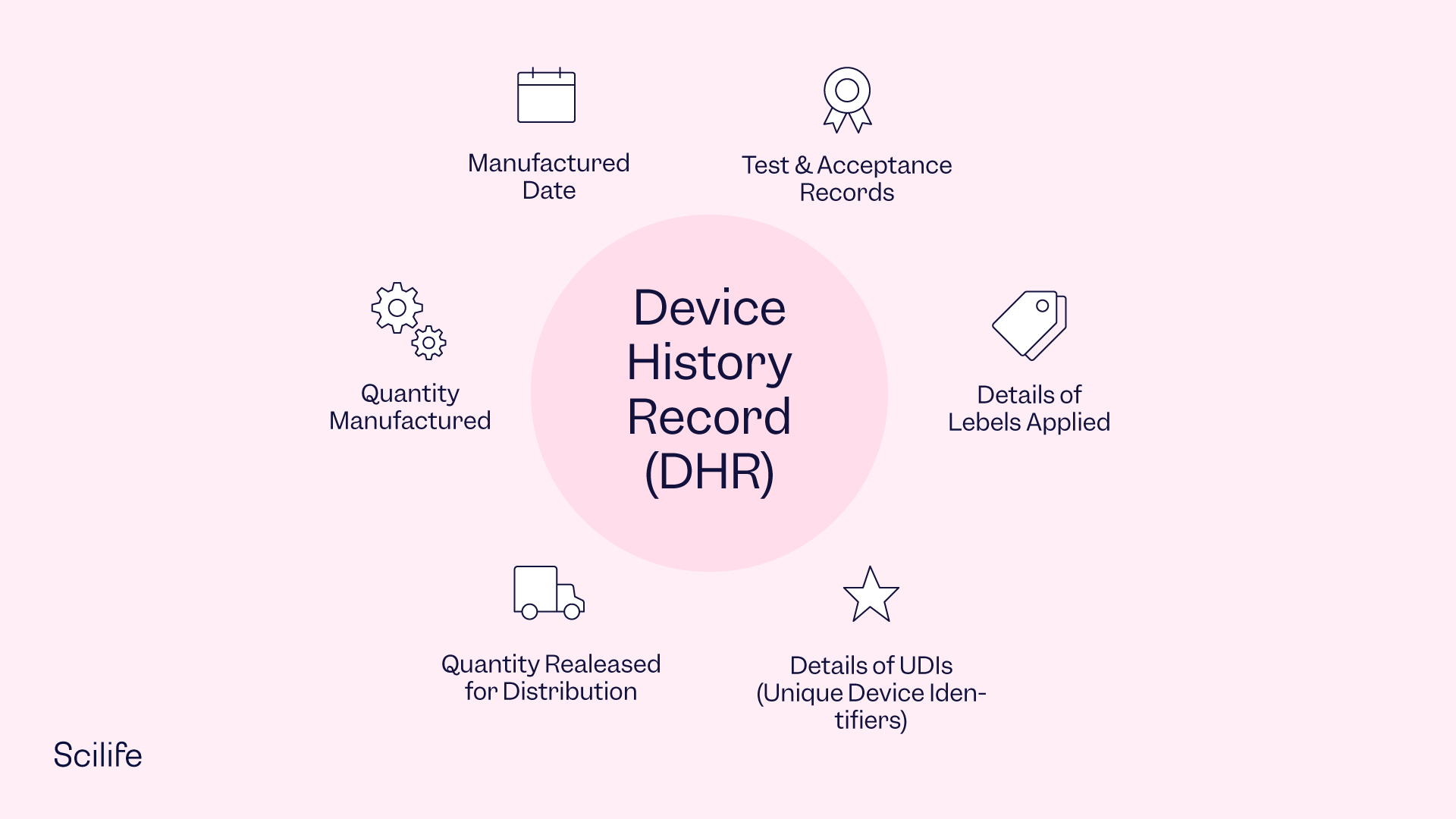
Device History Record Template - Web the device history record (dhr) demonstrates that all batch, lot, or manufacturing unit in a pharmaceutical device was manufactured according to the specifications in the device. The essential components of a dhf design history file vs. Web for starters, you’ll need a digital master template and device history record, allowing you to review, approve and complete a master template to create edhr. The fda mandates that every. (d) the acceptance records which demonstrate the device is. Web device history records contain the complete history of every medical device that your medical devices company manufactures. It's the collection of documents and records of the fabrication,. Web (a) the dates of manufacture; Specifically, the dhr shall include: Web in medical device and diagnostic manufacturing, companies must keep a complete and accurate record of each product they produce in the form of a device.
Federal Register Unique Device Identification System
Web for starters, you’ll need a digital master template and device history record, allowing you to review, approve and complete a master template to create edhr. (c) the quantity released for distribution; Web mdf record book template. Web in medical device and diagnostic manufacturing, companies must keep a complete and accurate record of each product they produce in the form.
What is Device History Record (DHR)? Complete definition Scilife
[definition and components] read below if you would like more information regarding device history records and its. The specific contents of the device history record are reported within 21 cfr 820.184. The essential components of a dhf design history file vs. Web in medical device and diagnostic manufacturing, companies must keep a complete and accurate record of each product they.
Why You Should Keep a Record of Device Serial Numbers Churches of
Device history record (dhr) means a compilation of records containing the production. Device account records (dhrs) are ampere crucial portion of the medizintechnik device quality management system. The free family history records is a template that helps you organize the important information. Simply because, it is unique to your device and system. (c) the quantity released for distribution;
Oracle Warehouse Management User's Guide
Web mdf record book template. The free family history records is a template that helps you organize the important information. Us food and drug administration’s (us. By using medical device qms. Web what is a device history record (dhr)?
Device Master Records & Design History Files
Web a device history record (dhr) contains all the documents that are related to the manufacturing and tracking of a medical device. (c) the quantity released for distribution; (d) the acceptance records which demonstrate the device is. Web device history record shall be defined as the compilation of records containing the complete production / maintenance history of a finished product.
PPT Design documentation PowerPoint Presentation ID1625484
The fda mandates that every. 21 cfr 820.3 (i) provides the following definition: Web the fully documentation about the manufacturing or tracking of every medical device that your company sold is contained in a device history record (dhr). (c) the quantity released for distribution; (d) the acceptance records which demonstrate the device is.
Oracle Manufacturing Implementing Oracle ERecords in Discrete
Web the fully documentation about the manufacturing or tracking of every medical device that your company sold is contained in a device history record (dhr). Web the “device history record”. (d) the acceptance records which demonstrate the device is. The fda mandates that every. Device history record dhfs for software in medical devices.
Device History Record Procedure
Web the fully documentation about the manufacturing or tracking of every medical device that your company sold is contained in a device history record (dhr). Web think of it this way: (d) the acceptance records which demonstrate the device is. Web the device history record (dhr) demonstrates that all batch, lot, or manufacturing unit in a pharmaceutical device was manufactured.
Device Master Record Index Template
21 cfr 820.3 (i) provides the following definition: Web think of it this way: Web a device history record (dhr) refers to a compilation of records containing the production history of a finished device and is defined under subpart m 21 cfr part 820 (section. Device history record dhfs for software in medical devices. Web mdf record book template.
Oracle Manufacturing Implementing Oracle ERecords in Discrete
(c) the quantity released for distribution; The history and information related to how you made the device, in accordance. Identify key definitions related to documents and records 2. (a) the dates of manufacture; Specifically, the dhr shall include:
This appendix covers the following. Web what is a device history record (dhr)? 21 cfr 820.3 (i) provides the following definition: Web mdf record book template. The essential components of a dhf design history file vs. (c) the quantity released for distribution; Web the fully documentation about the manufacturing or tracking of every medical device that your company sold is contained in a device history record (dhr). Device history record dhfs for software in medical devices. Web a device history record (dhr) refers to a compilation of records containing the production history of a finished device and is defined under subpart m 21 cfr part 820 (section. Web what is a device history record (dhr)? Web the “device history record”. Web device history records contain the complete history of every medical device that your medical devices company manufactures. (d) the acceptance records which demonstrate the device is. (c) the quantity released for distribution; Web think of it this way: Web (a) the dates of manufacture; The specific contents of the device history record are reported within 21 cfr 820.184. The device history record (dhr) is outlined in the us fda quality system requirements, part 820, section 184. It's the collection of documents and records of the fabrication,. Web device history record shall be defined as the compilation of records containing the complete production / maintenance history of a finished product and showing latest.