Iso 13485 Audit Checklist Template
Iso 13485 Audit Checklist Template - Web the checklist is created by reviewing the iso 13485:2016 standard and any documented procedures or undocumented processes for the activity to determine what should happen. Web march 29, 2021 in the iso 13485:2016 quality management system (qms), management of suppliers is a critical part of the success of the purchasing process. Web be ready for qms inspections with an iso 13485 audit checklist. The checklist is best used by trained and practicing auditors to evaluate or assess quality management systems requirements based on. Ad become iso certified with expert guidance. Ensure that you have a clear understanding of the requirements of iso 13485 to effectively use it, too. Web pdf template, an iso 13485 audit checklist is utilized by quality managers to determine if the organization’s qms is aligned with the iso 13485:2016 standard. Web the auditor will use pro qc’s standard audit checklist as a tool that thoroughly assesses the device manufacturer’s quality management system against the iso 13485 standard. Use this template print as pdf title page organization conducted on date prepared by. Oliver eidel the iso 13485 is the standard for quality management in the medical device industry.
Iso 13485 Internal Audit Schedule Template Gambaran
Familiarize yourself with the checklist to understand the questions or criteria it includes. Web looking to make your own or download iso 13485 audit checklist template to view all the tasks required and tick off the tasks when completed? Web pdf template, an iso 13485 audit checklist is utilized by quality managers to determine if the organization’s qms is aligned.
ISO 13485 2016 Internal Auditor Checklist TQS Inc.
Ensure that you have a clear understanding of the requirements of iso 13485 to effectively use it, too. Web forms and checklists are used to record data, capture information, and facilitate compliance with iso 13485 requirements. The iso 13485 audit checklist allows quality managers to document evidence of compliance based on processes, standard requirements, and process characteristics. Here are all.
Iso 13485 Internal Audit Checklist
Web the auditor will use pro qc’s standard audit checklist as a tool that thoroughly assesses the device manufacturer’s quality management system against the iso 13485 standard. The checklist is organized by the quality management subsystem. Web objective parties conduct internal audits. Web pdf template, an iso 13485 audit checklist is utilized by quality managers to determine if the organization’s.
Iso 13485 Audit Checklist 2016 doctorazgard
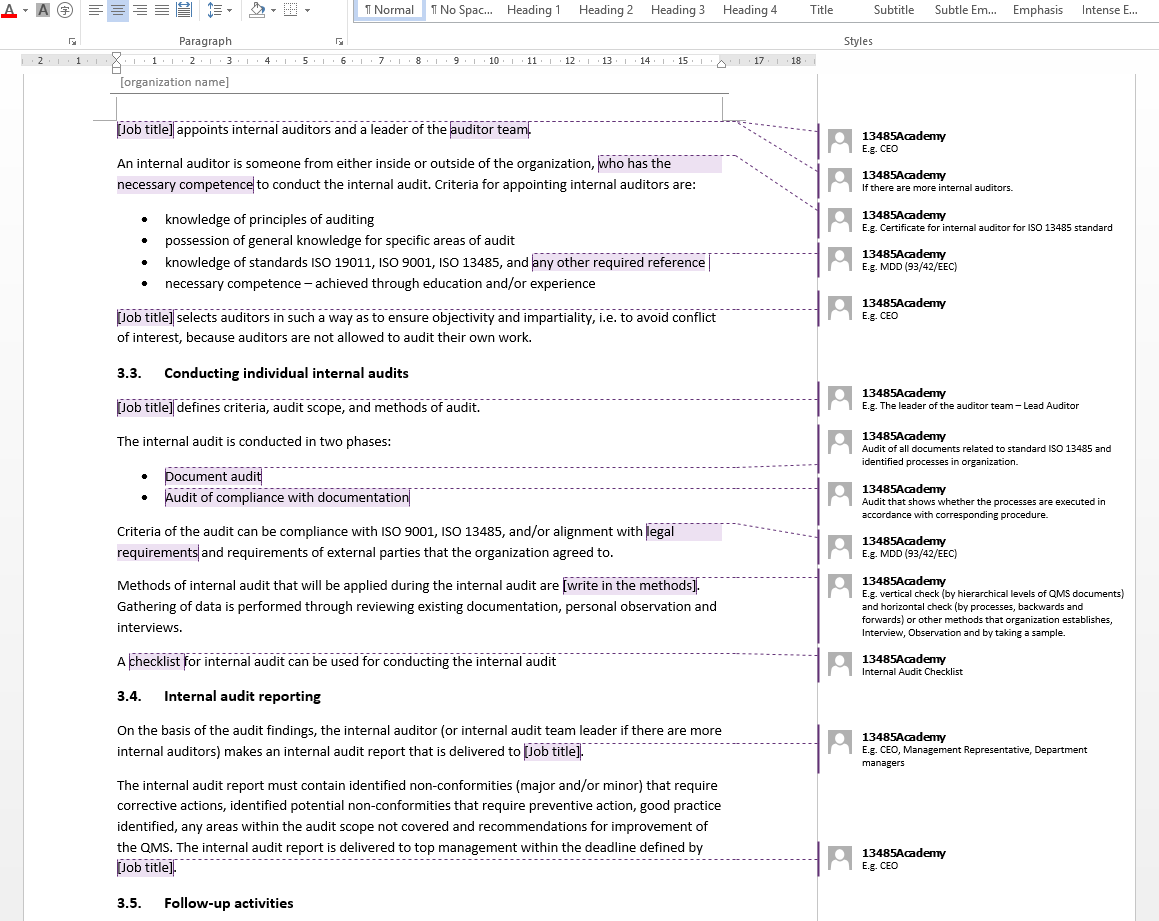
The documentation template may be used for iso 13485 certification audit purposes. Web july 25, 2022 iso 13485 templates dr. Web an iso 13485 audit checklist is used by quality managers to determine whether the company's quality management system (qms) is compliant with the iso 13485:2016 standard. Criteria for evaluation and selection of suppliers: Web be ready for qms inspections.
Iso 13485 Internal Audit Checklist
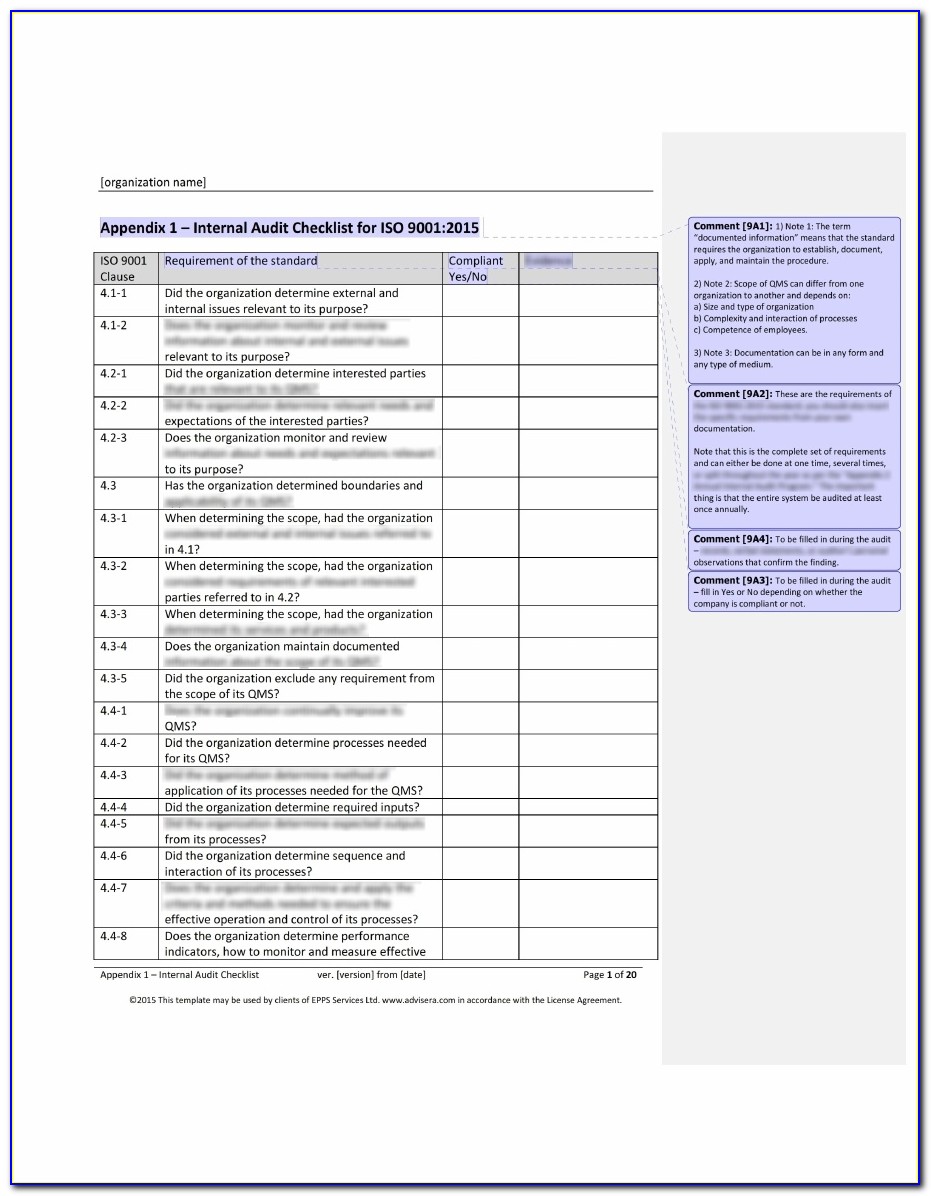
Web download a free audit plan template today to streamline auditing processes, stay compliant with safety standards, and improve operations. The checklist is best used by trained and practicing auditors to evaluate or assess quality management systems requirements based on. Document templates contain an average of twenty comments each, and offer clear guidance for filling them out. Ad become iso.
Iso 13485 internal audit checklist advfaher
Web looking to make your own or download iso 13485 audit checklist template to view all the tasks required and tick off the tasks when completed? Web an mdsap audit checklist is a tool used by quality managers to determine if the manufacturer’s qms meets the requirements of iso 13485:2016 and that of regulatory authorities participating in the mdsap. Iso.
ISO 13485 Quality Management and Document Control Software Internal
This digital checklist is divided into 5 sections following iso 13485: Web the documentation template may be used for iso 13485 certification audit purposes. Document templates contain an average of twenty comments each, and offer clear guidance for filling them out. Oliver eidel the iso 13485 is the standard for quality management in the medical device industry. Web an iso.
Iso 13485 internal audit checklist currentvast
It helps assess whether a company is ready to undergo an iso 13485:2016 certification audit by a third party. Web the documentation template may be used for iso 13485 certification audit purposes. Web the checklist is created by reviewing the iso 13485:2016 standard and any documented procedures or undocumented processes for the activity to determine what should happen. Web the.
Internal Audit Checklist Template Iso 13485
Web looking to make your own or download iso 13485 audit checklist template to view all the tasks required and tick off the tasks when completed? The iso 13485 audit checklist allows quality managers to document evidence of compliance based on processes, standard requirements, and process characteristics. Familiarize yourself with the checklist to understand the questions or criteria it includes..
Iso 13485 internal audit checklist hitswes
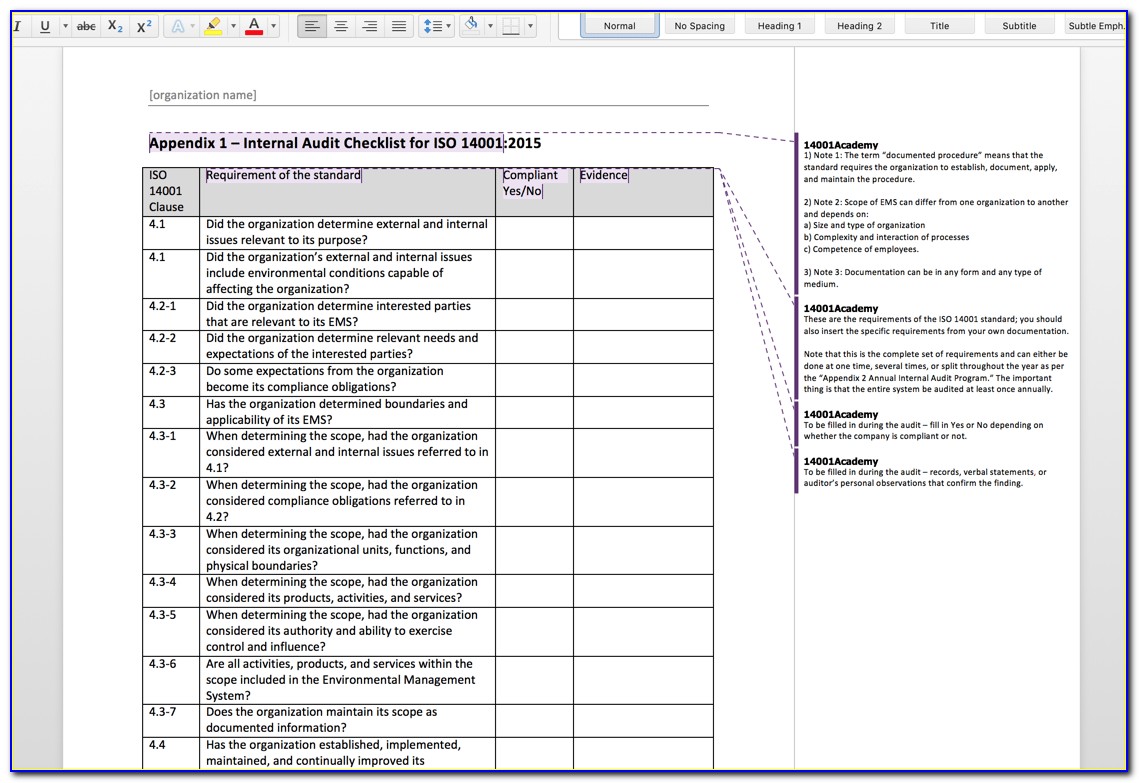
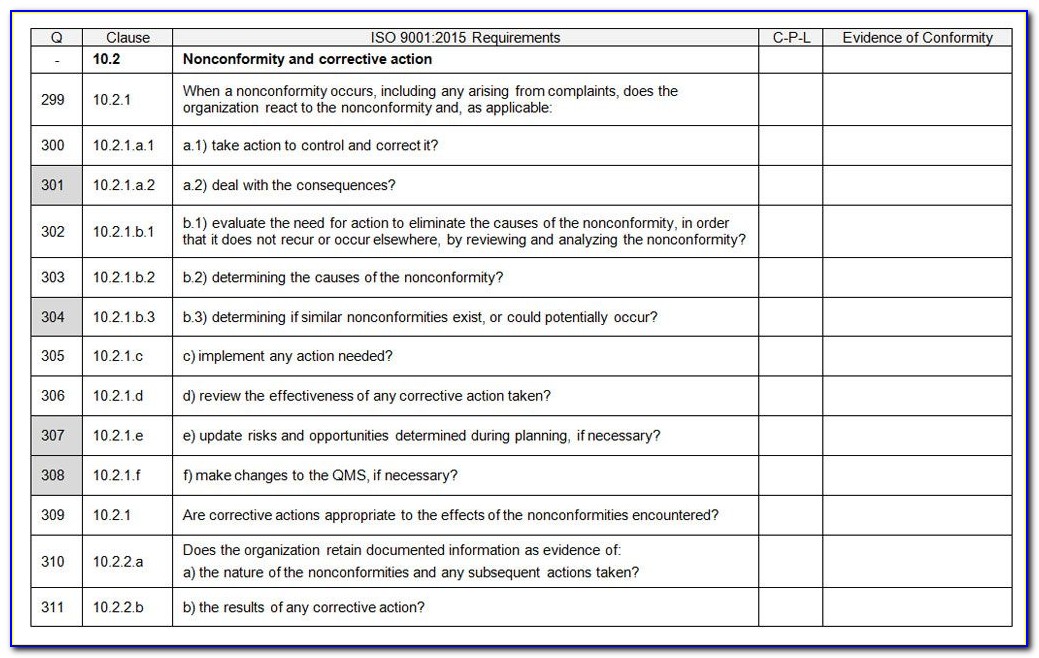
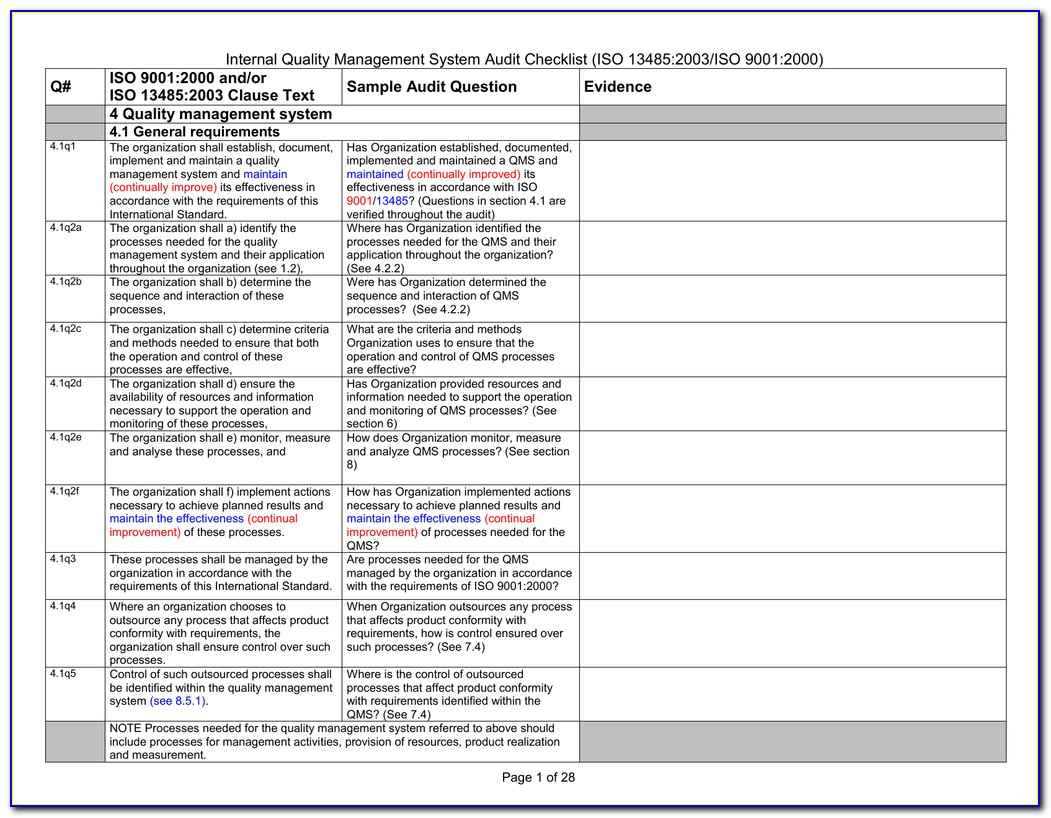
Web 4 quality management system 4.1 general requirements 4.1q1 the organization shall establish, document, implement and maintain a quality management system and maintain (continually improve) its effectiveness in accordance with the requirements of this international standard. Use this template print as pdf title page organization conducted on date prepared by. Web an iso 13485 audit checklist is used by quality.
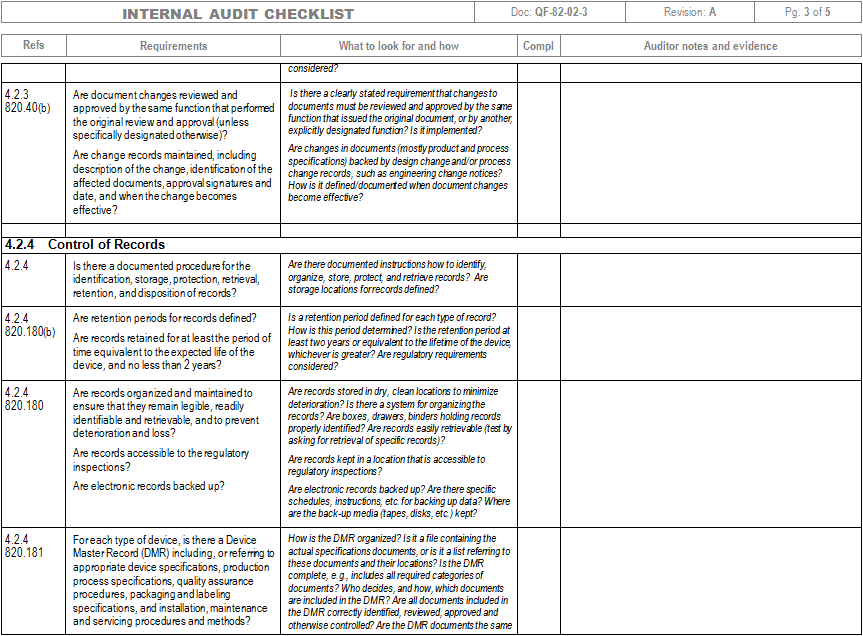
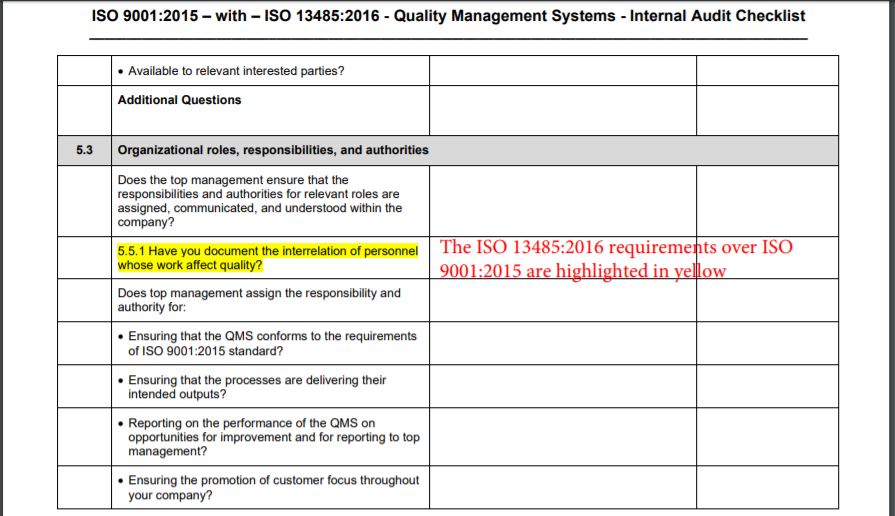
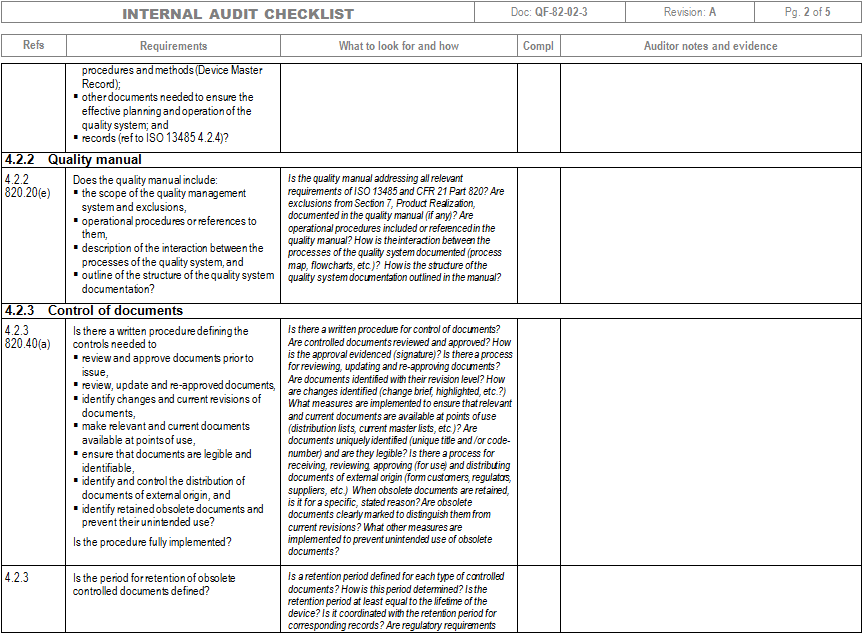
Web pdf template, an iso 13485 audit checklist is utilized by quality managers to determine if the organization’s qms is aligned with the iso 13485:2016 standard. Use an iso 13485:2016 checklist to help identify process gaps. The quality manual defines the scope of your qms and its procedures within your qms and describes the interaction of processes within your qms. Web an mdsap audit checklist is a tool used by quality managers to determine if the manufacturer’s qms meets the requirements of iso 13485:2016 and that of regulatory authorities participating in the mdsap. Web download an iso 13485 risk management plan template. Ad become iso certified with expert guidance. Web march 29, 2021 in the iso 13485:2016 quality management system (qms), management of suppliers is a critical part of the success of the purchasing process. The iso 13485 audit checklist allows quality managers to document evidence of compliance based on processes, standard requirements, and process characteristics. View or download free iso 13485 audit checklist to help you organise all the tasks from here. Web the documentation template may be used for iso 13485 certification audit purposes. Use this iso 13485 internal audit checklist template to determine whether the company's quality management system (qms) is compliant with the iso standards. Web an iso 13485 audit checklist is used by quality managers to determine whether the company's quality management system (qms) is compliant with the iso 13485:2016 standard. Web looking to make your own or download iso 13485 audit checklist template to view all the tasks required and tick off the tasks when completed? Web objective parties conduct internal audits. This digital checklist is divided into 5 sections following iso 13485: It helps assess whether a company is ready to undergo an iso 13485:2016 certification audit by a third party. The checklist is organized by the quality management subsystem. Web an iso 13485 audit checklist is utilized by quality managers to determine if the organization’s qms is aligned with the iso 13485:2016 standard. Web the auditor will use pro qc’s standard audit checklist as a tool that thoroughly assesses the device manufacturer’s quality management system against the iso 13485 standard. Use this template print as pdf title page organization conducted on date prepared by.