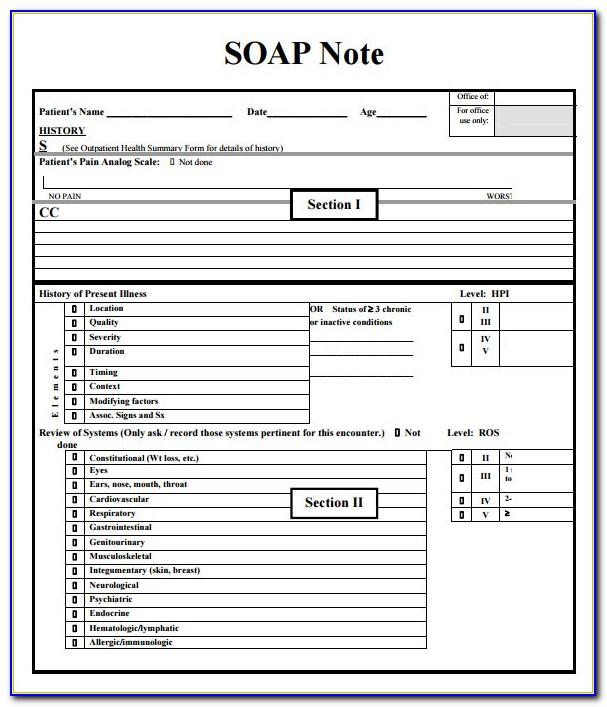
Note To File Template Clinical Research
Note To File Template Clinical Research - Text enclosed with <> is a placeholder for a specific detail (e.g.,. Web november 30, 2015 this post is unconventional and perhaps a little unpopular, but it has to be said: Web clinical study tools and templates. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many. Responsibility to be used by principal investigators and study team. Web the button contains templates, sample forms, guidelines, regulations and informational materials to supporting investigators at an company and conduct to high quality clinical. The primary purpose of every tmf is to tell the story of a. Regulatory binder pointer page templates; Example 1 (doc) example 2 (doc) example 3 (doc) Web valid notes to file (ntfs) should, at minimum, meet the following basic criteria:
2019 Pediatric Clinical Note Template2 Medical Diagnosis Health Care
Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by nih that are being conducted under a fda ind or ide application. Web research findings and/or have a delegate of the bcca vp of research sit in on research study meetings. It is used to clarify an error, omission or discrepancy.
Note To File Sample HQ Printable Documents
Kept on file in the site regulatory file and made available to the clinical site monitors reviewing the site’s. Web research findings and/or have a delegate of the bcca vp of research sit in on research study meetings. Web regulatory binder protocol deviation log template; The primary purpose of every tmf is to tell the story of a. Web this.
FREE 38+ Notes Samples in Google Docs MS Word Apple Pages
Web the button contains templates, sample forms, guidelines, regulations and informational materials to supporting investigators at an company and conduct to high quality clinical. Web welcome to global health trials' tools and templates library. Web valid notes to file (ntfs) should, at minimum, meet the following basic criteria: Web clinical study tools and templates. Web a note to the study.
PPT Orientation for New Clinical Research PERSONNEL Module 2
I will provide, every 6 months, a written summary regarding data. Responsibility to be used by principal investigators and study team. Web november 30, 2015 this post is unconventional and perhaps a little unpopular, but it has to be said: Kept on file in the site regulatory file and made available to the clinical site monitors reviewing the site’s. Web.
Clinical Trial Report Template (1) TEMPLATES EXAMPLE TEMPLATES
Web valid notes to file (ntfs) should, at minimum, meet the following basic criteria: Web the following page provides a template for the content and format of a note to the study file. Web the button contains templates, sample forms, guidelines, regulations and informational materials to supporting investigators at an company and conduct to high quality clinical. Example 1 (doc).
Clinical Progress Note Template
Web the button contains templates, sample forms, guidelines, regulations and informational materials to supporting investigators at an company and conduct to high quality clinical. Web valid notes to file (ntfs) should, at minimum, meet the following basic criteria: Web note to file examples: Web a note to the study file should be retained and stored, as follows: Web this clinical.
Clinical Progress Note Format
Include the subject and protocol to which it refers. Responsibility to be used by principal investigators and study team. Web a note to the study file should be retained and stored, as follows: Web healthcare business proposal templates. Web the following page provides a template for the content and format of a note to the study file.
Note To File Template Download by Pharma Student Issuu
Web healthcare business proposal templates. Documents are important and essential in the healthcare and medical industries. More than likely, you are writing and filing too many. Web the following page provides a template for the content and format of a note to the study file. Web the button contains templates, sample forms, guidelines, regulations and informational materials to supporting investigators.
181121 File Note template
Responsibility to be used by principal investigators and study team. This template assists the study team in contacting study participants. Web valid notes to file (ntfs) should, at minimum, meet the following basic criteria: Please note that this page has been updated for 2015 following a quality check and review of the templates, and many. Documents are important and essential.
Editable Clinical Progress Note Template Counseling Dap inside Dap Note
Responsibility to be used by principal investigators and study team. The primary purpose of every tmf is to tell the story of a. Web the following page provides a template for the content and format of a note to the study file. Include the subject and protocol to which it refers. This template assists the study team in contacting study.
Text enclosed with <> is a placeholder for a specific detail (e.g.,. Web this template provides a recommended structure for documenting note to files for research studies. Web research findings and/or have a delegate of the bcca vp of research sit in on research study meetings. I will provide, every 6 months, a written summary regarding data. The primary purpose of every tmf is to tell the story of a. Web nimh note to file (ntf) template version 1.0 nimh version 1.0 july 2019page 2of 2 nimh note t o file (ntf) template tool summary remove tool summary before. Web clinical study tools and templates. Web regulatory binder protocol deviation log template; Include the subject and protocol to which it refers. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many. Regulatory binder pointer page templates; Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by nih that are being conducted under a fda ind or ide application. Web november 30, 2015 this post is unconventional and perhaps a little unpopular, but it has to be said: The number of documents listed below includes. Web welcome to global health trials' tools and templates library. Web healthcare business proposal templates. More than likely, you are writing and filing too many. Responsibility to be used by principal investigators and study team. Web valid notes to file (ntfs) should, at minimum, meet the following basic criteria: Kept on file in the site regulatory file and made available to the clinical site monitors reviewing the site’s.