Regulatory Strategy Template For Medical Devices
Regulatory Strategy Template For Medical Devices - Class i, class ii or class iii, based on the level of control necessary to provide reasonable assurance of its safety and. Web each device is assigned to one of three regulatory classes: Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Web strategy and implementation plan. Web here are the guidelines: Department of health and human services, food and drug. These are the most important ones so you should probably get started. Web feb 18, 2021 strategy for regulatory compliance for mdr with template as european regulatory compliance becomes more complicated many medical device. The regulatory, quality, compliance and strategy experts your life science you need. I am looking for templates for us, canada and eu regulatory compliance for.
Regulatory Globe Gap Analysis Template Medical Devices, HD Png
Web a new requirement for a manufacturer of medical devices and in vitro diagnostics (ivds) is to have a strategy for regulatory compliance. Web here are the guidelines: Web feb 18, 2021 strategy for regulatory compliance for mdr with template as european regulatory compliance becomes more complicated many medical device. Ad click now to learn more about rca's global consulting.
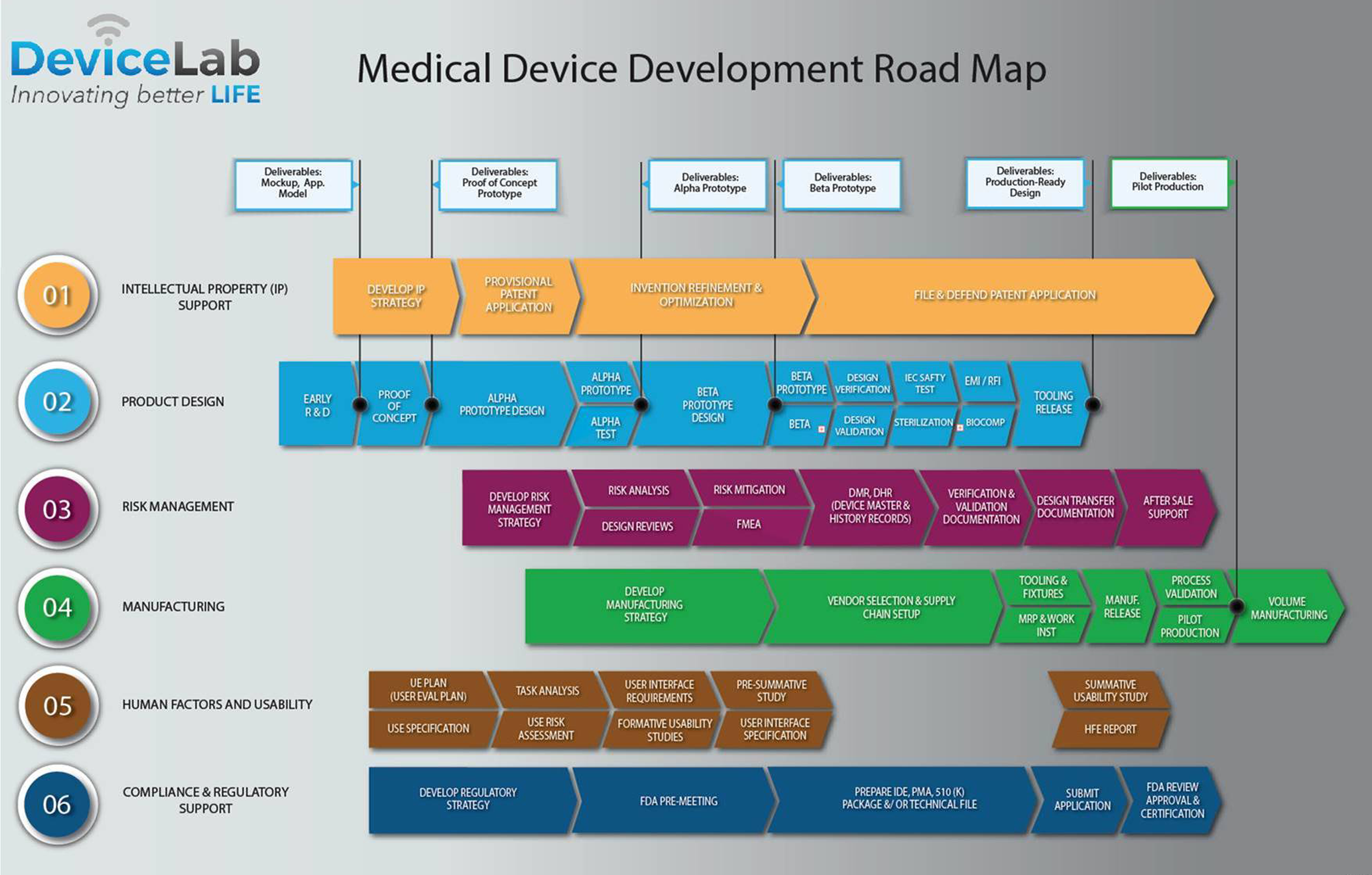
The Medical Device Development Process at DeviceLab Part 1
Class i, class ii or class iii, based on the level of control necessary to provide reasonable assurance of its safety and. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Contains nonbinding recommendations (version october 6, 2021) 2 Web feb 18, 2021 strategy for.
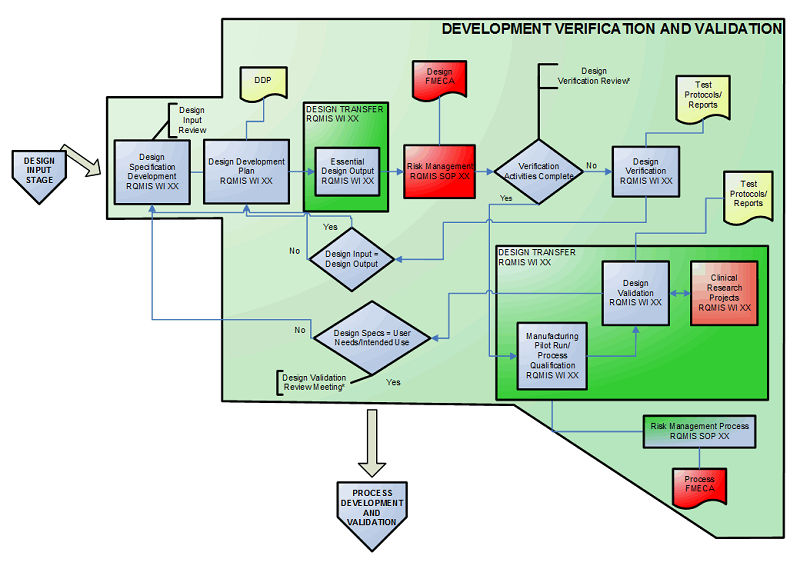
Fda Design Control Guidance Document
Determining a cost/return on investment for. Establish a robust medical device patient safety net in the united states 2. A regulatory strategy is more than just picking a. Contains nonbinding recommendations (version october 6, 2021) 2 Indications for use (ifu) your team should develop an ifu (a basic description of how the device is intended to be used), and should.
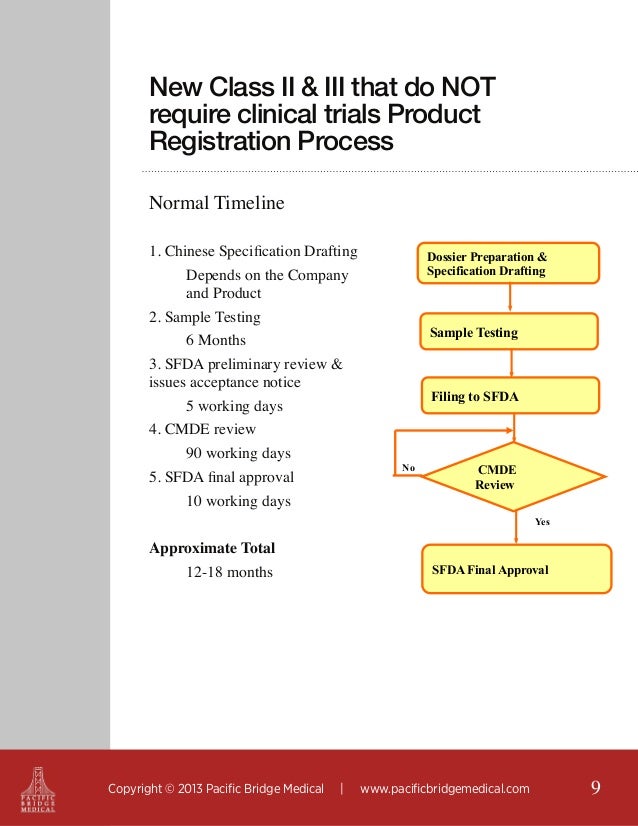
Medical Devices; US and Chinese legislation Kvalito
This comprises regulatory provisions like registration requirements and processes, but also (not legally binding) guidelines as well as predictive approval. Establish a robust medical device patient safety net in the united states 2. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Web these include.
Image result for design control phases medical device Medical device
This comprises regulatory provisions like registration requirements and processes, but also (not legally binding) guidelines as well as predictive approval. Class i, class ii or class iii, based on the level of control necessary to provide reasonable assurance of its safety and. Web a new requirement for a manufacturer of medical devices and in vitro diagnostics (ivds) is to have.
Is Regulatory compliance strategy for medical devices effective
Web background where does regulatory strategy fit in product development? Contains nonbinding recommendations (version october 6, 2021) 2 Web a new requirement for a manufacturer of medical devices and in vitro diagnostics (ivds) is to have a strategy for regulatory compliance. Web 1 additionally, to successfully navigate the complex regulatory system, recognize and respect factors such as development timelines, budgets,.
Regulatory Strategies for Medical Device Companies to Succeed in Asia
Web background where does regulatory strategy fit in product development? Contains nonbinding recommendations (version october 6, 2021) 2 Ad management consultants offering the world's best business toolkits, frameworks & templates. The regulatory, quality, compliance and strategy experts your life science you need. Indications for use (ifu) your team should develop an ifu (a basic description of how the device is.
Stringent Regulatory Authority The Regulation Of Wearable Medical
Web these include your intended use (super important) and your mdr classification document, among others. Ad management consultants offering the world's best business toolkits, frameworks & templates. Web planning your medical device global market regulatory strategy. Web when we are speaking about regulatory strategy for medical devices, three major strategies can cause delays in obtaining approval to market in any.
Regulatory Compliance printable pdf download
Web strategy and implementation plan. This comprises regulatory provisions like registration requirements and processes, but also (not legally binding) guidelines as well as predictive approval. Web a new requirement for a manufacturer of medical devices and in vitro diagnostics (ivds) is to have a strategy for regulatory compliance. Web please use one of the following formats to cite this article.
UK MHRA Guidance Medical devices EU regulations for MDR and IVDR
Web a new requirement for a manufacturer of medical devices and in vitro diagnostics (ivds) is to have a strategy for regulatory compliance. Web #1 does anyone have a regulatory plan template that they would like to share? Ad click now to learn more about rca's global consulting network and service solutions. The regulatory, quality, compliance and strategy experts your.
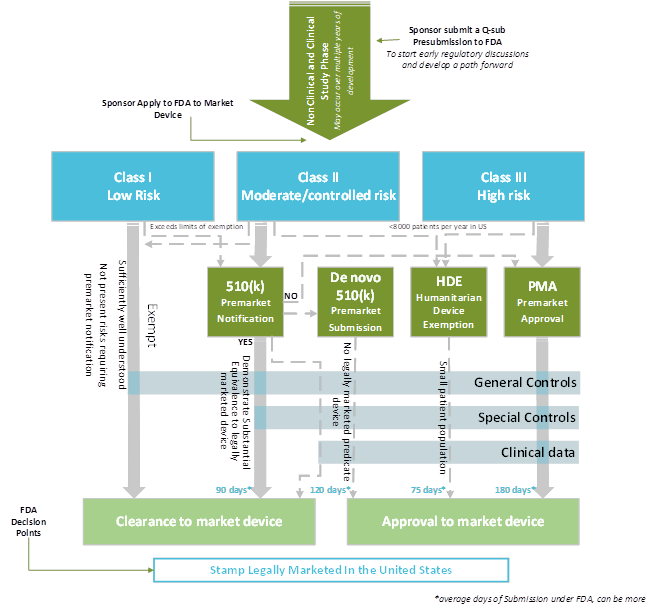
Web these include your intended use (super important) and your mdr classification document, among others. Web each device is assigned to one of three regulatory classes: Indications for use (ifu) your team should develop an ifu (a basic description of how the device is intended to be used), and should include:. Class i, class ii or class iii, based on the level of control necessary to provide reasonable assurance of its safety and. These are the most important ones so you should probably get started. Ad click now to learn more about rca's global consulting network and service solutions. Web background where does regulatory strategy fit in product development? Web an effective regulatory compliance strategy for medical devices must contain a number of elements, including: Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Ad management consultants offering the world's best business toolkits, frameworks & templates. Web when we are speaking about regulatory strategy for medical devices, three major strategies can cause delays in obtaining approval to market in any country. The regulatory, quality, compliance and strategy experts your life science you need. Web strategy and implementation plan. Web a new requirement for a manufacturer of medical devices and in vitro diagnostics (ivds) is to have a strategy for regulatory compliance. Contains nonbinding recommendations (version october 6, 2021) 2 Web please use one of the following formats to cite this article in your essay, paper or report: Establish a robust medical device patient safety net in the united states 2. I am looking for templates for us, canada and eu regulatory compliance for. A regulatory strategy is more than just picking a. Web feb 18, 2021 strategy for regulatory compliance for mdr with template as european regulatory compliance becomes more complicated many medical device.